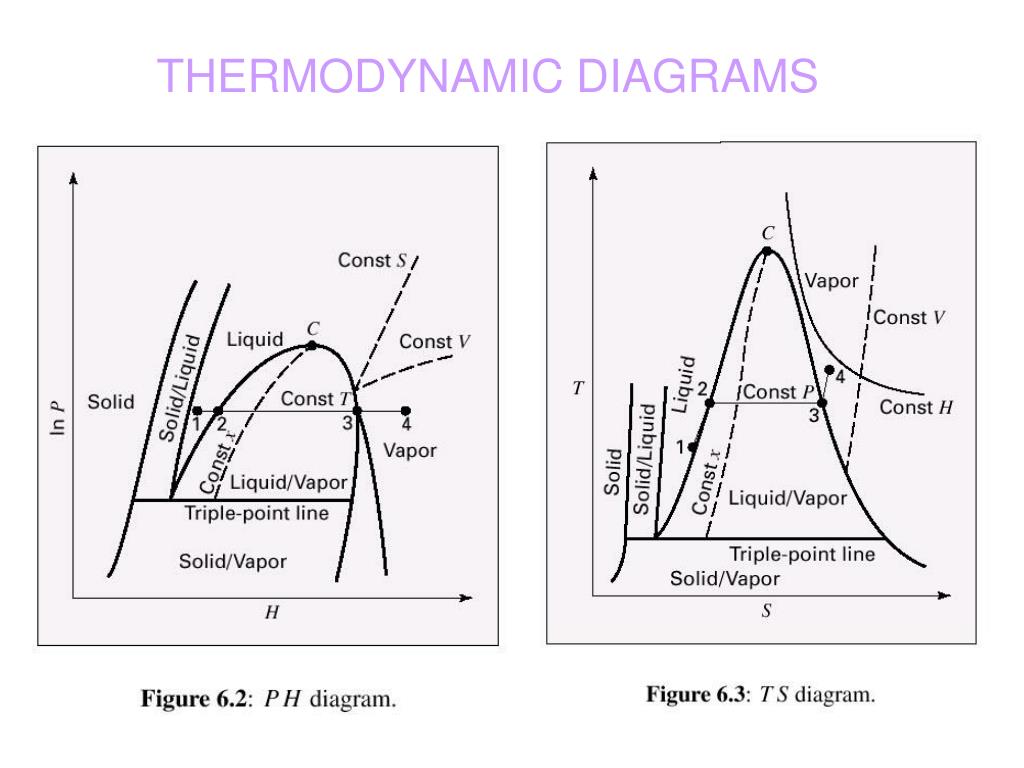
The temperature and its corresponding pressure at each point on the vaporization line are called saturation temperature, T_.įigure 2.3.4 T-v diagram Figure 2.3.5 P-v diagram 2.3.3 The saturated liquid-vapour two-phase regionįor a saturated liquid-vapour, two-phase mixture, we define quality x to denote the mass fraction of the saturated vapour in the mixture. Each point on the vaporization line represents an equilibrium state of saturation the substance is either a saturated liquid, a saturated vapour, or a two-phase liquid-vapour mixture. The curve that lies between the liquid and vapour phases is called vaporization line. In the P-T diagram, the two-phase regions appear as curves separating different single phases. The liquid and vapour phases are often called compressed liquid and superheated vapour, respectively. The single phase regions are labeled as solid, liquid, and vapour or gas in the P-T diagram. Thermo: Thermodynamics and Phase Equilibrium component of Chemical Engineering Design Library (ChEDL) Creating Objects Temperature-dependent Methods. When analyzing processes and cycles, these two-dimensional phase diagrams are commonly used, and therefore will be discussed in detail here.įigure 2.3.1 Pressure-specific volume-temperature (P-v-T) diagram 2.3.1 P-T diagramįigure 2.3.2 shows a generic pressure-temperature, P-T, diagram, from which we can observe three single-phase regions, three curves representing the two-phase mixtures, and two unique points: the triple point and the critical point. The three-dimensional P-v-T phase diagram can be projected to generate two-dimensional phase diagrams, such as P-T, P-v, and T-v diagrams. This phase diagram clearly shows the single phase regions of solid, liquid, and vapour or gas, as well as three two-phase regions, where solid-liquid, liquid-vapour, or solid-vapour coexist in equilibrium. Figure 2.3.1 is a three-dimensional P-v-T phase diagram, where the three axes are pressure, specific volume, and temperature, respectively. The equilibrium state of a pure substance and its phase transitions are commonly illustrated in phase diagrams. For example, liquid water at 1 atm turns into ice when its temperature drops to the freezing point of 0 oC. 
High entropy means high disorder and low energy. PV diagrams - part 2: Isothermal, isometric, adiabatic processes. PV diagrams - part 1: Work and isobaric processes. Scientists refer to the measure of randomness or disorder within a system as entropy. First law of thermodynamics problem solving. The more energy that is lost by a system to its surroundings, the less ordered and more random the system is. When its temperature or pressure changes, a substance may transition from one phase to another. An important concept in physical systems is that of order and disorder. 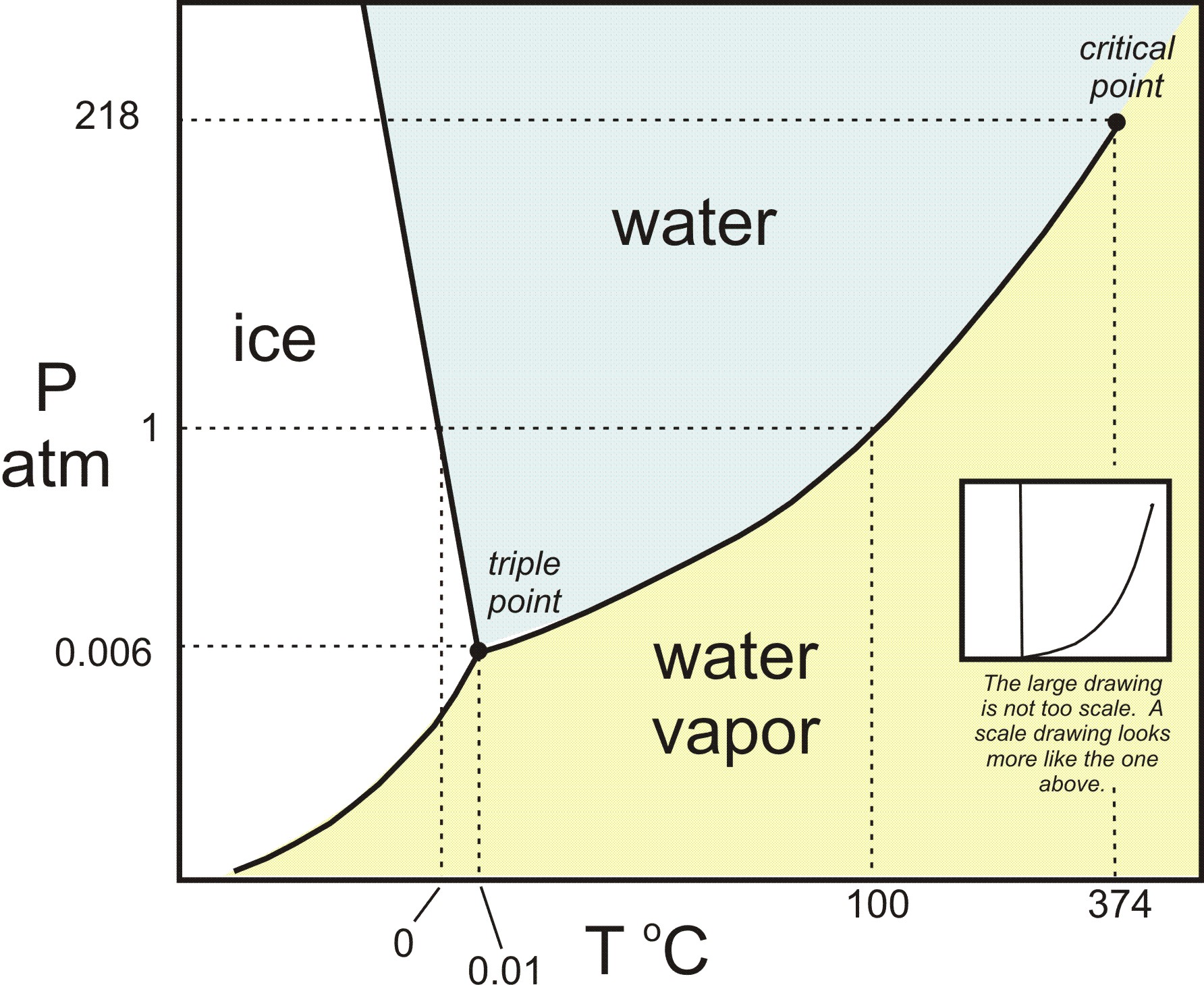
A pure substance may exist in any of the three phases: solid, liquid, and vapour, at certain temperatures and pressures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
